OAB Monograph
Overview of Overactive Bladder
OAB Description
Epidemiology and Relevant Risk Factors
Overactive bladder (OAB), characterized by frequent urinary urgency and nocturia, with or without urge incontinence,1 affects approximately 35 million* people in the US2,3 and significantly impairs activities of daily living (ADL). An internet-based survey study among 20,000 US adults ≥40 years found the overall prevalence of OAB to be 23%, with the prevalence of at least “sometimes” to be 27.2% and 43.1% and “often” to be 15.8% and 32.6%, in men and women respectively.2 The study found prevalence to increase with age, from 16.9% among men 40–45 years of age to 49.4% among older men ≥76 years of age and from 36.6% among women 40–45 years of age to 51.2% among older women ≥76 years of age.2
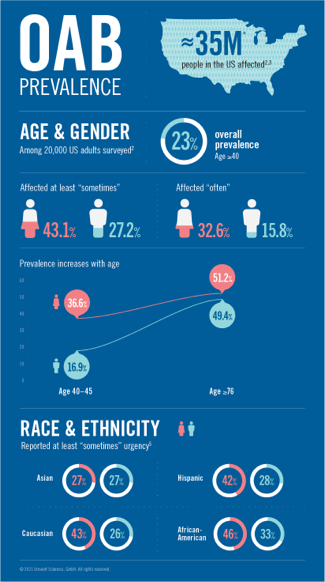
Apart from age and gender, studies have shown the risk of OAB to vary by race and ethnicity. The NHANES study4 found non-Hispanic Black and Mexican-American women to be 1.7 (95% CI: 1.4–1.8) times more likely to report urge incontinence compared to non-Hispanic White women. In contrast, the study by Coyne et al.5 found the prevalence of OAB of at least “sometimes” urgency to range from 27% in Asian women, 42% in Hispanic women, 43% in Caucasian women to 46% in African-American women. The study also found the prevalence of OAB to range from 26% in Caucasian men, 27% in Asian men, 28% in Hispanic men to 33% in African-American men. (Figure 1) Additionally, type 2 diabetes mellitus and metabolic syndrome are risk factors for developing OAB, while patients with OAB and comorbidities such as enlarged prostate are at increased risk of other disorders of bladder function. Many patients with cognitive decline, such as with stroke or Alzheimer’s disease, develop an overactive bladder with associated incontinence.6
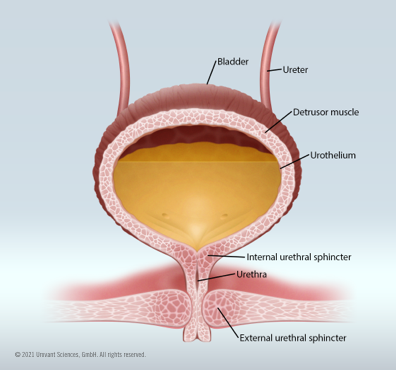
Female bladder filling (storage)
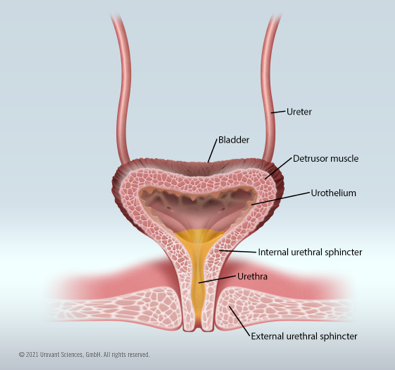
Micturition (voiding)
Pathophysiology
Neurologic innervation and bladder muscle control
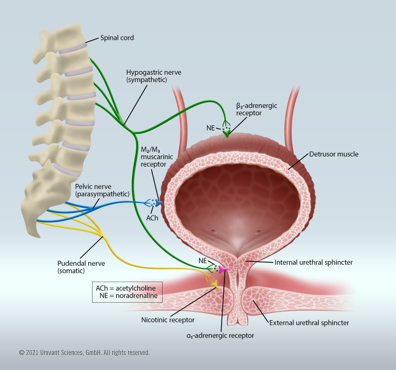
The process of micturition is a neural feedback process; as the bladder fills, stretch receptors send signals through a spinal pathway, which normally will trigger the detrusor muscle of the bladder to contract and the internal urethral sphincter to relax once a certain volume is reached. This results in an urge to urinate. This reflex is primarily governed by parasympathetic autonomic activation, while urine storage and retention are more associated with sympathetic drivers. Voluntarily controlled signals from the pontine micturition center (PMC) in the brainstem go through the spinal cord to signal the detrusor muscle to relax and the internal sphincter to constrict, thus preventing urine leakage and enabling regular bladder control. When it is time to urinate, the PMC signals the detrusor muscle to constrict and the internal urethral sphincter to relax, leading to voiding urine. Voluntary control of micturition is accomplished when the PMC signals the external urethral sphincter to constrict, and urine can be retained.
There are many causes of overactive bladder, but in general, OAB occurs when the detrusor muscle experiences frequent involuntary contractions associated with increased activation of the parasympathetic nervous system. This is modulated by heightened stimulation of muscarinic neuronal receptors and subsequent acetylcholine release from the parasympathetic nerve terminal. This causes relative overactivity of the detrusor muscle and the accompanying urinary urgency sensation.9
An appropriate balance between the activity of the parasympathetic and sympathetic arms of the autonomic nervous system are key to normal bladder function. For addressing the symptoms of OAB, the traditional treatment paradigm targets the parasympathetic processes; normally, parasympathetic activity is relatively inhibited during urine storage, resulting in relaxation of the detrusor muscle (via M3 muscarinic receptors) and contraction of the internal urethral sphincter. Thus, anti-muscarinic agents can be used to decrease the micturition reflex sensitivity. An alternative approach is to provide relative activation of the sympathetic arm by introducing β3-adrenergic receptor agonists. Since there are β3-adrenergic receptors expressed on the bladder detrusor, this approach can relax the muscle, allowing the bladder to fill more before the voiding sensation is triggered.8
Clinical Presentation
The International Continence Society (ICS) defines OAB as “urinary urgency, usually accompanied by increased daytime frequency and/or nocturia, with urinary incontinence (OAB-wet) or without (OAB-dry), in the absence of urinary tract infection or other detectable disease.”10 American Urological Association (AUA) guidelines concur that these symptoms of day- and night-time urinary frequency and urgency, with or without urgency incontinence, if self-reported as bothersome, may indicate an OAB diagnosis.1
AUA suggests that up to seven micturition episodes during daytime may be considered normal frequency, but the actual total can be ”highly variable based upon hours of sleep, fluid intake, comorbid medical conditions and other factors.”1 ICS defines urinary incontinence (UI) as “involuntary loss of urine during the bladder storage phase,” with two incontinence subtypes: urgency urinary incontinence (UUI), for which ICS defines urgency as the “sudden, compelling desire to pass urine which is difficult to defer” and stress urinary incontinence, defined as “involuntary loss of urine on effort or physical exertion including sporting activities or on coughing or sneezing.“ ICS explains nocturia as the interruption of sleep one or more times at night due to the need to void, with each urination being “followed by sleep or the intention to sleep,”10 OAB treatment trials often utilize total incontinence episodes as a response measure, regardless of incontinence subtype (urgency, stress, or mixed).1
Diagnostic Approach
According to the latest AUA guidelines, a diagnostic process should be initiated by documenting symptoms and signs characterizing OAB and ruling out other disorders associated with patients’ symptoms. In this process, minimum requirements are a careful history, physical exam and urinalysis. The guidelines recommend that when assessing the signs and symptoms of OAB, the clinician should consider factors such as hesitancy or straining to void, history of urinary retention, force and intermittency of stream, excessive and type of fluid intake (e.g., with and without caffeine), difficulty getting to the bathroom on time and mobility, and current medications, such as diuretics, that affect bladder function. The degree of bother associated with the symptoms and its impact on activities of daily living (ADL) should also be assessed.1
The presence of other conditions must be ruled out when diagnosing OAB. If OAB symptoms (e.g., frequency, urgency and urinary incontinence) occur only at night, then nocturnal polyuria (production of 20% to 33% or more [>20% and >33% for patients with <65 and ≥65 years of age, respectively] of total 24-hour urine output at night), low nocturnal bladder capacity, or both should be ruled out. Unlike the frequent small-volume voids associated with OAB, if urinary frequency is associated with normal or large volume voids that match the volume of intake, polydipsia (“excessive drinking”) should be ruled out. If voiding is frequent and exceeds intake volume, the rare condition of diabetes insipidus should also be ruled out. Absence of UTI and hematuria should be confirmed. Additionally, if symptoms of urinary frequency and urgency, with or without urgency incontinence, are present with bladder pain, pelvic pain (including dyspareunia), or both, the clinical presentation may indicate interstitial cystitis or bladder pain syndrome. Atrophic vaginitis, which is also associated with incontinence symptoms, should be considered when diagnosing OAB among females.1
The guidelines further suggest a directed physical examination that includes an abdominal exam to assess for issues indicative of urinary retention. For patients receiving more than behavioral therapy, post-void residual (PVR) assessment using ultrasound can be performed to validate an OAB diagnosis. Diagnosis guidelines also recommend a rectal/genitourinary exam to rule out pelvic floor problems in women and prostatic pathology concerns in men, and a urine culture to rule out infections.1
Bladder diaries, used by patients to record their intake and voiding behaviors, are a useful tool for clinicians in the diagnostic process, as well as for patients as they self-monitor symptom levels.1 European Association of Urology (EAU) guidelines recommend asking patients to complete a voiding diary for a duration of 3–7 days when standardized assessment is needed. EAU points out that such diaries can further be used to measure treatment response and are widely used in clinical trials.11
Results from symptom questionnaires such as Urogenital Distress Inventory (UDI)12, the UDI-6 Short Form13, the Incontinence Impact Questionnaire (II-Q)13 and the Overactive Bladder Questionnaire14 may also be obtained at clinicians’ discretion. A Mini-Mental State Examination (MMSE)15 is suggested for patients at risk for cognitive impairment to determine degree of risk with anticholinergic therapy and aggravation of symptoms associated with cognitive problems.1
Clinical and Economic Burden of OAB
Impact on Psychosocial Functioning and Quality of Life (QoL)
Multidimensional impact of overactive bladder (OAB) on quality of life (QoL)20
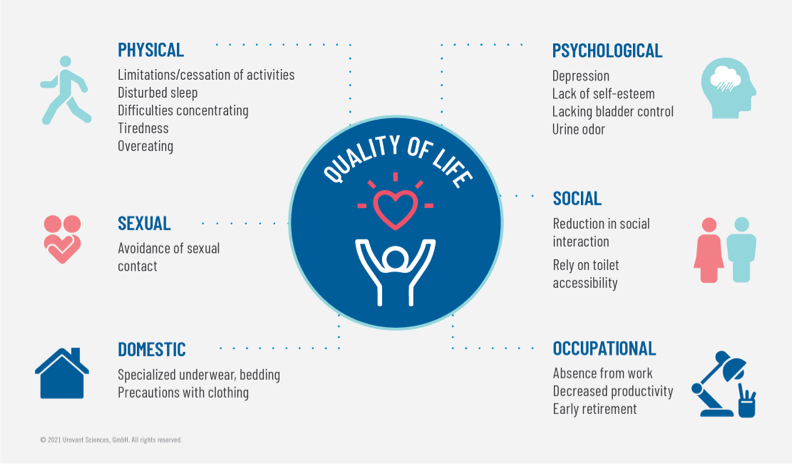
The negative impact of OAB on psychosocial functioning and QoL has been reported by several studies. In a population‐based, cross‐sectional, internet‐based survey by Sexton et al., using the Hospital Anxiety and Depression Score (HADS) instrument, the depression and anxiety rates (using a cut-off score of 8 for clinically relevant levels of both depression and anxiety separately) among those with OAB symptoms sometimes or more were 17.3% and 16.4% for men and 15.7% and 23.6% for women, respectively.16 In a 2001–2010 Medicare Current Beneficiary Survey, of adults ≥65 years old with scores ≥3 on the Vulnerable Elders Survey-13, Chuang et al.17 found a higher prevalence of depression among patients with OAB (defined as presence of an OAB diagnosis code or self-reported use of antimuscarinic medications) vs patients without OAB (23.9% vs 11.7%; p< .01). In the same study, patients with OAB were more likely to report ADL limitations (OR: 1.4 [95% CI: 1.1–1.7]), instrumental ADL limitations (OR: 1.5 [95% CI: 1.2–2.0]), physical functioning limitations (OR: 1.9 [95% CI: 1.0–3.5]) and depression (OR: 2.2 [95% CI: 1.8–2.8]), compared to patients without OAB. Similarly, in a study using Medicare Current Beneficiary Surveys from 2006 to 2011, Ganz et al.18 found that patients with OAB reported high levels of functional impairment as measured by the ADL (44% vs 33%), Instrumental ADL (53% vs 40%), and physical functioning limitation scales (90% vs 81%), compared to patients without OAB. In a survey study of 51 adults with OAB by Lai et al.19, the prevalence of depression (HADS ≥8) and moderate to severe depression (HADS-D ≥11) among patients with OAB were 27.5% and 12.0%, respectively. The authors found that patients with OAB reported significantly higher HADS depression scores compared to age-matched controls (5.3 ± 3.9 vs 2.8 ± 3.9, p= .004) and that patients with OAB with depression reported more severe incontinence symptoms as measured by International Consultation on Incontinence – Urinary Incontinence Short Form (ICIQ-UI; p= .001), greater bother (p= .01) and more impact on QoL as measured by Urogenital Distress Inventory Short Form (UDI-6; p< .001) andIncontinence Impact Questionnaire Short Form (IIQ-7; p< .001).
In a national telephone survey study by Stewart and colleagues,21 OAB was associated with significantly lower physical and mental component scores on the 36-Item Short Form Survey (SF-36)22, higher scores on the Center for Epidemiologic Studies Depression Scale23, and poorer scores on the Medical Outcomes Study (MOS) Sleep Scale24, which achieve statistical significance at all measures. In a population-based internet survey study by Milsom et al., physical and mental component scores on the SF-12 questionnaire were significantly lower among patients with OAB vs those with no/minimal symptoms of OAB, and for patients with OAB with or without bother (p< .001). The proportion of patients with an overall health rating of “excellent” was greater among those with no/minimal symptoms vs those with OAB with or without bother (no/minimal symptoms: 18.5% vs OAB with bother: 4.5% or OAB without bother: 8.0%; p< .001). The proportion of patients with a HADS score of ≥8 (clinically relevant levels of anxiety and depression, respectively) was significantly greater among patients with OAB with or without bother vs those with no/minimal OAB symptoms (OAB with bother: 43.5% and 36.5%, OAB without bother: 26.0% and 18.0%, with no/minimal symptoms: 13.5% and 9%, for anxiety and depression, respectively; p< .001).25 In a review of 32 studies, Kinsey et al. found patients with OAB were likely to have greater levels of depression, anxiety and embarrassment/shame; difficulties with social life; poor sleep quality and reported psychological impact on sexual relationships; and a lower QoL than people without OAB.26 Kannan et al.,27 using the National Health and Wellness Survey, found patients with OAB/urinary incontinence symptoms to have 4.1 points lower SF-8 mental and physical health scores, compared to patients without OAB/urinary incontinence symptoms (p< .001).
Impact on Work Productivity
OAB symptoms, including incontinence, have been shown to be associated with reduced work productivity, excess days related to absenteeism, disability and higher unemployment rates. In a population-based internet-based survey study by Sexton et al.,28 a higher proportion of patients with OAB reported having to always worry about interrupting meetings with frequent trips to the bathroom, compared to patients without OAB (35% vs 4%; p< .0001). In a US study using administrative claims data, Wu et al.29 reported employees with OAB had 2.2 excess days related to medically-related absenteeism and 3.4 excess days related to disability compared with patients without OAB (p< .01). In an internet-based survey conducted by Coyne et al.,30 patients with OAB vs those with no or minimal OAB symptoms were significantly more likely to be unemployed (44% men, 54% women vs 24% men, 41% women) and have greater productivity loss (work productivity and activity percent impairment: 19% men, 21% women vs 4% men, 7% women). Tang et al., using data from Adelphi OAB/UI disease-specific program, reported that patients with OAB with incontinence were less likely to be on paid employment (31.2% vs 42.3%; p< .001) and reported greater productivity loss (mean Work Productivity and Activity Impairment (WPAI) score: 31 vs 22; p< .001) compared to those without incontinence.31 Kannan et al.27 reported patients with OAB/urinary incontinence symptoms to have 8.0% greater overall work productivity loss (p< .001) and 12.9% greater activity impairment (p< .001), compared to patients without OAB/urinary incontinence symptoms.
Clinical Burden
OAB is associated with significant clinical burden as evidenced by a high prevalence of psychiatric comorbidities, UTIs and falls/fractures among patients with OAB. Sexton et al.16 found in a survey of respondents with OAB that 16% and 20% suffered from depression and anxiety, respectively. In another study, Chuang et al.17 found that 24% of OAB patients suffered from depression. Studies have shown that nearly half of patients with OAB develop urinary tract infections compared to patients without OAB (Chuang et al.17 reported 46% vs 16% and Yehoshua et al.32 reported 51% vs 28%, respectively). Onukwugha et al.33 estimated 48% of patients with OAB suffer from skin infections. Based on a review by Szabo et al., the proportion of patients who experience a fall over a 1-year period ranged from 19% to 50%.34
Economic Burden
Healthcare Costs
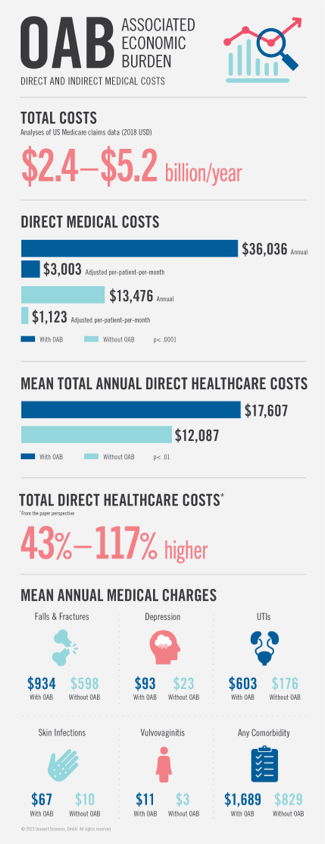
OAB has been shown to be associated with substantial economic burden in terms of direct and indirect medical costs. In an analyses of Medicare claims data, Puckrein et al.35 estimated the total US national costs associated with OAB to be $2.4 to $5.2 billion (2018 USD) per year. In a study by Durden et al.,36 using the MarketScan database, the annual and adjusted per-patient-per-month direct medical costs were estimated to be $36,036 and $3,003 for patients with OAB vs $13,476 and $1,123 for patients without OAB (p< .0001), respectively; whereas in a study by Chuang et al.,17 mean total annual direct healthcare costs for patients with OAB vs without OAB were $17,607 vs $12,087, respectively (p< .01). In a review of 18 studies, Powell et al. found the mean all-cause total direct healthcare costs (from the payer perspective) to range from $8,168 to $15,569 and OAB-specific costs to range from $656 to $860. Costs for patients with OAB were higher by 43% to 117%, compared to patients without OAB.37 Darkow et al.,38 in a retrospective claims database analyses, found mean annual medical charges to be significantly higher for patients with OAB vs patients without OAB, for falls and fractures ($934 vs $598; p< .0001); depression ($93 vs $23; p< .0001); UTIs ($603 vs $176; p< .0001); skin infections ($67 vs $10; p= .002); vulvovaginitis ($11 vs $3; p< .0001) and any comorbidity ($1,689 vs $829; p< .0001). (Figure 2)
...urinary incontinence was the most common health-related issue in people admitted from home to a long-term care facility (43%)...
Healthcare Resource Utilization

In the study by Chuang et al.,17 the proportion of patients with OAB vs without OAB that were institutionalized was 17.5% vs 10%, had hospital admissions was 37.5% vs 25.9%, outpatient visits was 86% vs 74%, physician office visits was 98% vs 92% and ER visits was 46.7% vs 33.7%, respectively, over a 12-month period (p< .01). Kannan et al.27 found patients with OAB/urinary incontinence symptoms to be 1.57 (95% CI: 1.47–1.68) times more likely to have emergency visits, 1.56 (95% CI: 1.43–1.69) times more likely to have hospitalization and 1.52 (95% CI: 1.41–1.63) times more likely to have medical provider visits, compared to patients without OAB/urinary incontinence symptoms. In a study by Thom et al.,40 a 50% and 30% higher risk of hospitalization was found among men and women ≥65 years of age with incontinence, respectively, compared to those without incontinence. The adjusted risk of admission to a nursing facility was 2.0 times greater for women with incontinence (95% CI: 1.7–2.4) and 3.2 times greater for men with incontinence (95% CI: 2.7–3.8), compared to those without incontinence. Holup et al.,41 in a study using the Minimum Data Set (MDS) for all free-standing, certified nursing facilities, among 46,695 subjects ≥65 years of age, found that urinary incontinence was the most common health-related issue in people admitted from home to a long-term care facility (43%), followed by dementia (38%) and a fall within 180 days (34%). Gorina et al.,42 using the long-term care MDS, found 70% of long-term care residents to have urinary incontinence.
Falls/Fractures-Related Healthcare Resource Utilization and Costs
OAB in elderly patients is associated with an increased risk of falls and fractures, resulting in increased healthcare resource utilization and costs. Caplan et al.,43 in a study among Medicare enrollees, aged 65 to 89 years, with osteoporosis, found 17.7% vs 14.9% (p< .001) of patients with vs without OAB to have a falls/fractures-related visit over a 12-month period. All-cause healthcare costs for patients with OAB were higher by 35% (p< .001), compared to patients without OAB. In another study among Medicare enrollees, aged 65 to 89 years, with dementia, Caplan et al.44 found 14.8% vs 11.9% (p< .001) of patients with vs without OAB to have a falls/fractures-related visit over a 12-month period, and all-cause healthcare costs for patients with OAB were higher by 23% (p< .001), compared to patients without OAB.
Use of anticholinergics and anticholinergic burden, particularly among older patients, have been increasingly reported in independent studies and analyses to be associated with cognitive impairment, including problems with memory.45 Anticholinergics used to treat OAB have also been associated with increased risk of falls and fractures.46,47 Inappropriate use of anticholinergics has also been associated with increased healthcare costs. Szabo et al.48 found an amplified risk of falls and fractures among OAB patients with higher vs no anticholinergic burden in the previous six months (HR: 1.4 [95% CI: 1.3–1.4]). A study by Yehoshua et al.32 found patients with OAB continuing to experience incontinence after initiating anticholinergic therapy were at a high risk for falls/fractures (46% higher adjusted risk; p< .001) and used significantly more healthcare resources (20% higher adjusted risk for hospital admissions; p= .012 and 33% higher adjusted total healthcare costs; p< .001) compared to patients without OAB. Suehs et al.49 studied the healthcare costs among elderly patients with OAB initiating anticholinergics and found annual all-cause healthcare costs to be significantly higher among those with vs without potentially inappropriate anticholinergic use ($12,001 [95% CI: $11,657–$12,355] vs $9,373 [95% CI: $9,113–$9,641]; p< .001).
Long-Term Care: Healthcare Resource Utilization and Costs
With an estimated prevalence of 70.3% among elderly patients in long-term care (LTC) facilities,42 OAB is associated with substantial healthcare resource utilization and costs in this patient population. In a study by Yeaw et al., using IQVIA’s LTC pharmacy data linked to IQVIA’s integrated data warehouse (Dx/Rx), the percentage of patients with outpatient visits was significantly higher in the OAB cohort compared to non-OAB cohort (physician office visits: 71.3% vs 65.3%; UTI-related visits: 33.2% vs 21.1%; urinary catheterization-related visits: 58.8% vs 39.6% and falls/fractures-related visits: 30.5% vs 25.5%) and patients with OAB incurred an adjusted 1.2 times (p< .0001) the total all-cause healthcare costs of patients in the matched non-OAB cohort (mean age 75 years). Unadjusted mean total healthcare costs for patients in the OAB cohort vs in the matched non-OAB cohort were $15,870 vs $12,975, respectively (p< .0001). Additionally, the authors found an adjusted 23% decrease in nonpharmacy costs, with mean total physician office costs reported as $1,152 vs $1,479, for treated vs untreated patients with OAB.50
References
- Gormley EA, Lightner DJ, Burgio KL, et al. Diagnosis and treatment of non-neurogenic overactive bladder (OAB) in adults: an AUA/SUFU guideline (2019). American Urological Association. Available at: https://www.auanet.org/guidelines/overactive-bladder-(oab)-guideline. Accessed June 09, 2020.
-
Coyne KS, Sexton CC, Vats V, Thompson C, Kopp ZS, Milsom I. National community prevalence of overactive bladder in the United States stratified by sex and age. Urology. 2011;77(5):1081-1087.
-
US Census Bureau. Age and Sex Composition: 2018. Available at: https://data.census.gov/cedsci/table?q=people%20by%20age%20group&t=Age%20and%20Sex&tid=ACSST1Y2018.S0101&hidePreview=false. Accessed November 4, 2020.
-
Dooley Y, Kenton K, Cao G, et al. Urinary incontinence prevalence: results from the National Health and Nutrition Examination Survey. J Urol. 2008;179(2):656-661.
-
Coyne KS, Margolis MK, Kopp ZS, Kaplan SA. Racial differences in the prevalence of overactive bladder in the United States from the epidemiology of LUTS (EpiLUTS) study. Urology. 2012;79(1):95-101.
-
Mayo Clinic. Overactive bladder: symptoms and causes [Mayo Clinic website]. Available at: https://www.mayoclinic.org/diseases-conditions/overactive-bladder/symptoms-causes/syc-20355715. Accessed August 18, 2020.
-
Fowler CJ, Griffiths D, de Groat WC. The neural control of micturition. Nat Rev Neurosci. 2008;9(6):453-466.
-
Verhamme KM, Sturkenboom MC, Stricker BH, Bosch R. Drug-induced urinary retention: incidence, management and prevention. Drug Saf. 2008;31(5):373-388.
-
Ellsworth PI, Kim ED. Overactive bladder [WebMD Medscape website]. January 27, 2020. Available at: https://emedicine.medscape.com/article/459340-overview#a5. Accessed November 4, 2020.
-
International Continence Society. ICS glossary of terminology [ICS website]. Available at: https://www.ics.org/glossary. Accessed January 8, 2021.
-
European Association of Urology. EAU non-oncology guidelines; urinary incontinence: 3.3 voiding diaries [EAU website]. Available at: https://uroweb.org/guideline/urinary-incontinence/#3. Accessed January 8, 2021.
-
Shumaker SA, Wyman JF, Uebersax JS, McClish D, Fantl JA. Health-related quality of life measures for women with urinary incontinence: the Incontinence Impact Questionnaire and the Urogenital Distress Inventory. Continence Program in Women (CPW) Research Group. Qual Life Res. 1994;3(5):291-306.
-
Uebersax JS, Wyman JF, Shumaker SA, McClish DK. Short forms to assess life quality and symptom distress for urinary incontinence in women: the Incontinence Impact Questionnaire and the Urogenital Distress Inventory. Neurourol Urodyn. 1995;14(2):131-139.
-
Coyne KS, Matza LS, Thompson C, Jumadilova Z, Bavendam T. The responsiveness of the OAB-q among OAB patient subgroups. Neurourol Urodyn. 2007;26(2):196-203.
-
Folstein MF, Robins LN, Helzer JE. The mini-mental state examination. Arch Gen Psychiatry. 1983;40(7):812.
-
Sexton CC, Coyne KS, Thompson C, et al. Prevalence and effect on health-related quality of life of overactive bladder in older Americans: results from the Epidemiology of Lower Urinary Tract Symptoms Study. J Am Geriatr Soc. 2011;59:1465-1470.
-
Chuang CC, Yang E, Zou KH, Araiza A, Wang A, Luo X. Health and functional status, health events, use of healthcare services and costs associated with overactive bladder among the medically complex vulnerable elderly in the US. Int J Clin Pract. 2019;73(8):e13207.
-
Ganz ML, Liu J, Zou KH, et al. Real-world characteristics of elderly patients with overactive bladder in the United States. Curr Med Res Opin. 2016;32(12):1997-2005.
-
Lai HH, Shen B, Rawal A, Vetter J. The relationship between depression and overactive bladder/urinary incontinence symptoms in the clinical OAB population. BMC Urol. 2016;16:60.
-
Tubaro, A. Defining overactive bladder: epidemiology and burden of disease. Urology. 2004:64(6 Suppl 1):2-6.
-
Stewart W, Van Rooyen J, Cundiff G, et al. Prevalence and burden of overactive bladder in the United States. World J Urol. 2003;20(6):327-336.
-
Ware J, Kosinski M, Keller S. SF-36 Physical and Mental Health Summary Scales: A User’s Manual. Boston, Mass: Health Assessment Lab, New England Medical Center; 1994.
-
Eaton WW, Smith C, Ybarra M, Muntaner C, Tien A. Center for Epidemiologic Studies Depression Scale: review and revision (CESD and CESD-R). In: Maruish ME, ed. The Use of Psychological Testing for Treatment Planning and Outcomes Assessment: Instruments for Adults. NJ: Lawrence Erlbaum Associates Publishers; 2004:363–377.
-
Allen RP, Kosinski M, Hill-Zabala CE, Calloway MO. Psychometric evaluation and tests of validity of the Medical Outcomes Study 12-item Sleep Scale (MOS sleep). Sleep Med. 2009;10(5):531-539.
-
Milsom I, Kaplan SA, Coyne KS, Sexton CC, Kopp ZS. Effect of bothersome overactive bladder symptoms on health-related quality of life, anxiety, depression, and treatment seeking in the United States: results from EpiLUTS. Urology. 2012;80(1):90-96.
-
Kinsey D, Pretorius S, Glover L, Alexander T. The psychological impact of overactive bladder: a systematic review. J Health Psychol. 2016;21(1):69-81.
-
Kannan H, Radican L, Turpin RS, Bolge SC. Burden of illness associated with lower urinary tract symptoms including overactive bladder/urinary incontinence. Urology. 2009;74(1):34-38.
-
Sexton CC, Coyne KS, Vats V, Kopp ZS, Irwin DE, Wagner TH. Impact of overactive bladder on work productivity in the United States: results from EpiLUTS. Am J Manag Care. 2009;15(4 Suppl):S98-S107.
-
Wu EQ, Birnbaum H, Marynchenko M, Mareva M, Williamson T, Mallett D. Employees with overactive bladder: work loss burden. J Occup Environ Med. 2005;47(5):439-446.
-
Coyne KS, Sexton CC, Thompson CL, et al. Impact of overactive bladder on work productivity. Urology. 2012;80(1):97-103.
-
Tang DH, Colayco DC, Khalaf KM, et al. Impact of urinary incontinence on healthcare resource utilization, health-related quality of life and productivity in patients with overactive bladder. BJU Int. 2014;113(3):484-491.
-
Yehoshua A, Chancellor M, Vasavada S, et al. Health resource utilization and cost for patients with incontinent overactive bladder treated with anticholinergics. J Manag Care Spec Pharm. 2016;22(4):406-413.
-
Onukwugha E, Zuckerman I, McNally D, Coyne K, Vats V, Mullins C. The total economic burden of overactive bladder in the United States: a disease-specific approach. Am J Manag Care. 2009;15(4 Suppl):S90-S97.
-
Szabo SM, Gooch KL, Walker DR, Johnston KM, Wagg AS. The association between overactive bladder and falls and fractures: a systematic review. Adv Ther. 2018;35(11):1831-1841.
-
Puckrein G, Walker D, Xu L, Congdon P, Gooch K. The prevalence and forecast prevalence of overactive bladder in the Medicare population. Clin Med Insights Urol. 2019;12. doi:10.1177/1179561119847464
-
Durden E, Walker D, Gray S, Fowler R, Juneau P, Gooch K. The direct and indirect costs associated with overactive bladder within a commercially-insured population in the United States. J Occup Environ Med. 2018;60(9):847-852.
-
Powell LC, Szabo SM, Walker D, Gooch K. The economic burden of overactive bladder in the United States: a systematic literature review. Neurourol Urodyn. 2018;37(4):1241-1249.
-
Darkow T, Fontes CL, Williamson TE. Costs associated with the management of overactive bladder and related comorbidities. Pharmacotherapy. 2005;25(4):511-519.
-
U.S. Centers for Medicare & Medicaid Services. MDS 3.0 Frequency Report: Fourth Quarter 2019; H0300: Bladder and Bowel – Urinary Incontinence. Available at https://www.cms.gov/Research-Statistics-Data-and-Systems/Computer-Data-and-Systems/Minimum-Data-Set-3-0-Public-Reports/Minimum-Data-Set-3-0-Frequency-Report. Accessed July 13, 2020.
-
Thom DH, Haan MN, Van Den Eeden SK. Medically recognized urinary incontinence and risks of hospitalization, nursing home admission and mortality. Age Ageing. 1997;26(5):367-374.
-
Holup AA, Hyer K, Meng H, Volicer L. Profile of nursing home residents admitted directly from home. J Am Med Dir Assoc. 2017;18(2):131-137.
-
Gorina Y, Schappert S, Bercovitz A, et al. Prevalence of incontinence among older Americans. Vital Health Stat 3. 2014;(36):1-33.
-
Caplan EO, Abbass IM, Suehs BT, et al. Impact of coexisting overactive bladder in Medicare patients with osteoporosis. Arch Gerontol Geriatr. 2018;75:44-50.
-
Caplan EO, Abbass IM, Suehs BT, Ng DB, Gooch K, van Amerongen D. Impact of coexisting overactive bladder in Medicare patients with dementia on clinical and economic outcomes. Am J Alzheimers Dis Other Demen. 2019;34(7-8):492-499.
-
Andre L, Gallini A, Montastruc F, et al. Association between anticholinergic (atropinic) drug exposure and cognitive function in longitudinal studies among individuals over 50 years old: a systematic review. Eur J Clin Pharmacol. 2019;75:1631-1644.
-
Marcum ZA, Wirtz HS, Pettinger M, et al. Anticholinergic medication use and falls in postmenopausal women: findings from the women’s health initiative cohort study. BMC Geriatr. 2016;16(1):76.
-
Zia A, Kamaruzzaman S, Myint PK, Tan MP. Anticholinergic burden is associated with recurrent and injurious falls in older individuals. Maturitas. 2016;84:32-37.
-
Szabo SM, Gooch K, Schermer C, et al. Association between cumulative anticholinergic burden and falls and fractures in patients with overactive bladder: US-based retrospective cohort study. BMJ Open. 2019;9:e026391. doi:10.1136/bmjopen-2018-026391
-
Suehs BT, Davis C, Franks B, et al. Effect of potentially inappropriate use of antimuscarinic medications on healthcare use and cost in individuals with overactive bladder. J Am Geriatr Soc. 2016;64(4):779-787.
-
Yeaw J, Shah D, Kincaid A, Mudd P. PUK5 Overactive bladder in the United States long-term care setting is associated with significant burden and increased healthcare resource utilization and costs: a retrospective database study. Value in Health. 2020;23:S377-S378.


